Back to basics: Medical gas storage under NFPA 99
The purpose of this article is to provide health care facility professionals with instructional guidance to help them reach NFPA 99 compliance associated with the storage of medical gases. Readers are advised to consult the NFPA 99 text itself, and this article will help with the initial understanding of the requirements.
Learning Objectives
- Learn about what NFPA 99 covers for medical gas storage requirements.
- Differentiate between the different types of medical gas storage rooms and their requirements.
- Know the common NFPA 99 compliance issues for medical gas storage rooms.
Medical gas system insights
- Bottles, manifolds, compressor and pump locations all have unique and detailed requirements for storage and housing within health care facilities.
- NFPA 99 provides specific guidelines to those designing and operating health care facilities that require medical gas.
At times, health care facilities use spaces to store medical gases — medical oxygen, nitrogen, nitrous oxide and others — that are noncompliant based on the requirements in the 2021 edition of NFPA 99: Health Care Facilities Code. This code specifies many health care facility requirements including operational, mechanical, electrical and architectural for the storage of such medical gases.
Misses on the code typically occur when closets, storage rooms and other spaces are repurposed for the storage of these gases. As a result, challenges for facility managers include:
- Unable to create designated spaces for medical gases.
- Efforts to maintain compliance become inconsistent.
- NFPA 99 requirements can be misinterpreted.
- Requirements may not be known.
Medical gas definitions NFPA 99
The following are critical definitions from NFPA 99-2021 to review before we go further.
- Container: A low-pressure, vacuum insulated vessel containing gases in liquid form.
- Cryogenic fluid central supply system: At a health care facility, an equipment assembly supplies compressed gas with a cryogenic fluid that terminates at the source valve.
- Cylinder: A supply tank for high-pressure gases with pressures that may exceed 13.8 kilopascals (kPa) or 2,000 pounds per square inch (psi) gauge.
- Medical gas: See medical support gas or patient medical gas below.
- Medical support gas: Nitrogen or instrument air used for any medical support purpose (e.g., to remove excess moisture from instruments, to operate medical-surgical tools). This gas may be used in laboratories and is not respired as part of any treatment. It falls under the general requirements for medical gases.
- Manifold: A device for connecting the outlets of one or more gas cylinders to the central piping system for that specific gas.
- Nonflammable: Not readily capable of burning with a flame and not liable to ignite and burn when exposed to flame.
- Oxidizing gas: A gas that supports combustion.
- Patient medical gas: Piped gases — such as oxygen, nitrous oxide, helium, carbon dioxide and medical air — that are used in the application of or calibration of medical devices for human respiration.
How medical gas storage rooms are used
Medical gas storage rooms are used to store frequently used medical gases. These include but are not limited to: oxidizing gases, such as oxygen and nitrous oxide, and inert gases, such as nitrogen, carbon dioxide and medical air. The use of medical gas storage rooms is regulated by NFPA 99 (along with other codes), which establishes the requirements for the design, construction, installation and use of medical gas storage rooms in health care facilities.
In addition to supplying health care practitioners with essential resources, gas storage rooms allow for safety in the storage and fulfillment of these gases. Although not covered in this article, medical gas storage compliance also requires an in-depth hazard analysis which is outlined in NFPA 55: Compressed Gases and Cryogenic Fluids Codes, 2020 edition. These analyses take such subjects as flammability, toxicity and explosivity of the gases into account.
- Reference Table 6.2.1 in NFPA 55 for the design and number of control areas per floor.
- Reference Table 6.3.1.1 in NFPA 55 for a list of all gases and the maximum allowable quantity (MAQ) of all varieties of gases within different sizes and types of control areas.
There are several types of gas storage rooms and each has specific characteristics as well as design and operational requirements depending on the type of gases being stored. First, gas storage rooms are characterized as either indoor rooms — a room within the interior of the health care facility itself — or outdoor areas — an outdoor enclosure that meets gas storage requirements.
Secondly, gas storage rooms are either with a central supply — a system for piping medical gases to different functional areas within the health care facility — or without a central supply, in which gases are stored independently and retrieved for use in other areas.
Requirements for all types of storage: All storage rooms (except for rooms with storage only of less than 300 cubic feet of nonflammable gases at standard temperature and pressure, known as STP) — whether they feature central supply systems or not — have shared requirements in terms of signage, temperature limits, accessibility, finishes and restraints.
Signage: For example, source locations containing both inert gases and positive pressure gases other than oxygen and medical air must have their doors labeled as follows (see Figure1):

Figure 1: Example storage rooms. Courtesy: Dewberry
Meanwhile, source locations containing only oxygen or medical air must have their doors labeled as follows:
Medical Gases
No Smoking or Open Flame
Proximity: In terms of proximity, full containers and cylinders must be segregated from all others (empty cylinders, etc.) — a commonly missed requirement, especially considering the constant activity of medical practice.
High-temperature limit: All areas with cylinders, whether they have central supply or not, must not exceed 125°F.
Finishes and racks: The finishes of the interior spaces or the exterior enclosures must be made of noncombustible materials (interiors can be limited-combustible materials), as well as any racks/shelves/supports for cylinders must be made of noncombustible materials (or limited-combustible materials).
Other: In general, these areas should have access to move cylinders with hand trucks, have lockable doors or gates and have proper restraints to prevent the cylinders from falling (whether full or empty). Other requirements are exclusion of: any fuel-fired equipment, heating elements greater than 266oF, flammable gas, flammable liquid or flammable vapors.
Best practice: It is best to use oxygen sensors and alarms in gas storage rooms to detect any harmful leaks and maintain 3 feet of clearance within each storage room around cylinders/containers and walkways for easy movement of cylinders. Neither of these suggestions is explicitly outlined in the language of NFPA 99.
Requirements for storage with a central supply included
A central supply system can be composed entirely of manifolds or the system may be a combination of manifolds and stored cylinders or containers. These systems can be located either within a single room in the health care facility (interior) or in an outdoor enclosure (exterior).
- Interior: Interior central supply systems locations are permitted to store manifolds for gas cylinders, manifolds for cryogenic liquid containers, in-building emergency reserves, instrument air standby headers, individual components on the oxygen side of the concentrator sources. Design and construction of interior central supply rooms, excluding cryogenic fluid center supply systems, have specific requirements outlined in NFPA 99. To highlight one of the indoor requirements specifically, rooms containing oxygen, nitrous oxide or other oxidizers shall be separated from the rest of the building by walls or floors with one-hour fire ratings and with doors and other openings with a ¾-hour rating. These rooms also require emergency power and specific ventilation which includes low-wall intakes (see Figure 2).

Figure 2: Interior storage with central supply for carbon dioxide. Courtesy: Dewberry
- Exterior: Exterior central supply system locations are permitted to store manifolds for gas cylinders, manifolds for liquid cryogenic containers, cryogenic fluid central supply and individual components on the oxygen side of the concentrator sources together. Exterior storage locations with central supply have less stringent, but unique requirements. Some of these unique requirements are protection from prolonged contact with moisture and soil, well drained area and special considerations when associated with natural elements (e.g., exposure to the sun) (see Figure 3).

Figure 3: Exterior storage with central supply for nitrogen. Courtesy: Dewberry
- Important limitations: The following central supply systems are not permitted to be located within any of the interior or exterior storage rooms with central supply mentioned in the sections above: medical air central compressor supply sources, medical-surgical vacuum central supply sources, waste anesthetic gas disposal central supply sources, instrument air compressor central supply sources, any other compressors, vacuum pump or electrically powered machinery and compressors, dryers and air receivers used to supply oxygen concentrators.
Requirements for storage without a central supply included
Any storage that does not include a central supply system will feature cylinders or containers that are not in use. Similar to the central supply systems, these can be located either within the interior of the health care facility or in an outdoor enclosure.
Interior: Interior storage rooms have unique requirements based on the volume of gases stored within those rooms. Specifically, storage spaces with fewer than 300 cubic feet of nonflammable gases (at STP), 300 to 3,000 cubic feet of nonflammable gases (at STP) or greater than 3,000 cubic feet of nonflammable gases (at STP) will differ in requirements for the ventilation, electrical and architectural features. Rooms with greater than 3,000 cubic feet of nonflammable gases (at STP) require walls or floors with one-hour fire ratings and with doors and other openings with a ¾-hour rating. Rooms with greater than 300 cubic feet of nonflammable gases (at STP) require specific ventilation which includes low-wall intakes (see Figure 4).

Figure 4: Interior storage only for medical air. Courtesy: Dewberry
Exterior: Exterior storage locations without central supply have less stringent requirements. One item to note is specific requirements for location as it relates to building exits, wall openings, intakes, property lines and combustibles (see Figure 5).

Figure 5: Exterior storage only for nitrogen and nitrous oxide. Courtesy: Dewberry
Although there are many similarities in the requirements of the different room types and number of gases stored, health care facility professionals need to understand the differences and limitations to each to confirm that their facility is constructed and operated within the NFPA 99 requirements. A matrix to review your facility’s compliance with NFPA 99 can be helpful (see Figure 6).
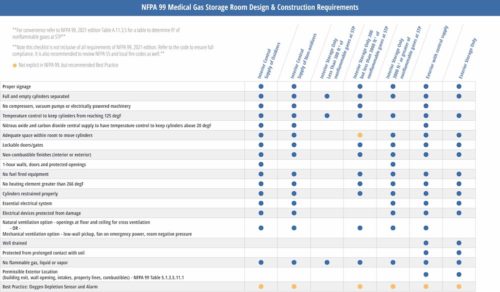
Figure 6: NFPA 99 medical gas storage room design and construction requirements matrix. Courtesy:
Dewberry
For user convenience, NFPA 99 outlines the typical medical gas cylinder’s volume and weight at STP for common cylinders seen in health care facilities (example: B, D, E, H) in the appendix under Table A.11.3.5. This table provides an easy way to determine how much gas is stored in each type of cylinder when evaluating spaces with various cylinder sizes. It is also common for cylinders to list the content’s total cubic feet at STP on the exterior of the cylinder.
What types of medical gas storage room problems exist?
Medical facilities that do not meet NFPA 99’s medical gas storage room requirements often end up that way unintentionally. Specifically, requirements may not be completely understood or health care staff may place medical gas cylinders in a room not specifically designed for that use. In doing so, facilities may inadvertently be out of compliance and at risk for penalties, citations and even human injury or death.
Fortunately, facility professionals can develop a better understanding of the code to keep their facilities in compliance. Additionally, they can become aware of some of the most common misinterpretations and address them directly. The following list consists of some common NFPA 99 storage room errors that the authors have seen in the field.
Rooms are misapplied or not understood: The NFPA 99 requirements for medical gas storage are different depending on the type of room involved. Additionally, noncompliant storage rooms are often used for the storage of medical gases without taking compliance into consideration. It’s important to fully understand the type of room in consideration and be familiar with the NFPA 99 requirements for that room type based on the descriptions above.
Rooms do not have correct features, such as electrical, signage or exhaust/ventilation: In addition to the signage requirements described above that are often missed, most storage rooms require ventilation. This ventilation can be achieved with natural ventilation, but typically (due to building layout) is mechanical ventilation. This requires low-wall exhaust, negative pressure and a means of makeup air. Mechanical ventilation exhaust is required to be on the facility’s essential electrical system as well. Natural ventilation requirements are in NFPA 99 Chapter 9.3.6.5.2 and requirements for mechanical ventilation can be found in Chapter 9.3.6.5.3. Rooms are often observed without exhaust systems or not meeting the specific requirements of these code sections. Emergency electrical provisions is also a requirement on most of the room types that is frequently missed.
The quantity of gas or number of containers exceeds the limits of the rooms: NFPA 99 has specific requirements regarding the amount of gas that can be stored in a room, with respect to less than 300 cubic feet of nonflammable gases at STP, between 300 cubic feet and 3,000 cubic feet of nonflammable gasses at STP and greater than 3,000 cubic feet of nonflammable gasses. It is critical not to exceed these thresholds. Often, it is missed that a room is designed for a certain threshold and is exceeding that threshold with the total cubic feet of stored gases in the room (not counting empty cylinder’s volume).
Manifolds or cryogenics are located too close to equipment in violation of requirements: Manifolds and cryogenics have unique requirements in terms of their proximity to other containers. If located outdoors, they must be installed in an enclosure used only for the enclosure of those containers and they must comply with minimum distance requirements. If located indoors, they must be installed within a room used only for those types of containers.
Design or construction does not include appropriate fire-rated walls and doors: As noted above, NFPA 99 requires that storage rooms with central supply of oxidizers and storage rooms without central supply and greater than 3,000 cubic feet of nonflammable gases at STP must be separated from the rest of the building by walls and floors having a one-hour fire resistance rating. They must also feature doors and other opening protectives having a ¾-hour fire protection rating. This is often missed when medical gases are placed in a room without proper design.
Outdoor storage is too close to building exits or air intakes: NFPA 99 requires that outdoor storage areas for medical gases must be located at a specific distance from building exits and air intakes. This is to prevent the spread of gas should a leak occur.
Medical gases are not stored behind locked doors: NFPA 99 requires that storage areas for medical gases must be locked when not in use. This is to prevent unauthorized access to the gases.
Medical gases lack restraints or fastening against tipping or falling: NFPA 99 requires that containers of medical gases are restrained or fastened against tipping or falling. This is to prevent accidents or leaks should a container fall over.
Incompatible central supply systems stored together: As mention in a previous section, NFPA 99 outlines central supply systems that can and cannot be stored together. This precaution prevents hazardous mistakes and potential risks if gases were to mix with machinery-based equipment, etc.
Facilities and respiratory staff fail to communicate on appropriate storage: Facility professionals must provide respiratory staff with appropriate guidance as they access and use medical gases. Respiratory staff may not understand the quantity limits of a certain room based on the design and construction of the room.
Other medical gas regulations
Health care facility professionals should also take note of other regulations that may apply to medical gas storage practices based on the type, location or region of facilities, as well as any risks associated with those aspects (e.g., higher likelihood of natural disasters).
For example, NFPA 55 has additional limitations on hazardous materials per control area. It includes limitations on the number of control areas per floor that should be observed in accordance with NFPA 99 requirements. Similarly, the local fire codes should be referenced when looking for additional limitations on storage quantities. Typically, the local fire codes expand upon the limitations outlined within NFPA 99 and NFPA 55.
Additionally, facility professionals should understand the code edition(s) currently enforced for their facility and study each edition. This article is written based on the 2021 edition of NFPA 99, which may have different requirements than what is enforced at a particular facility.
Additionally, if a storage room was constructed under a different code edition and was compliant at the time of construction, it is allowed to remain as it was originally constructed as long as the local authority having jurisdiction has determined the system is not a hazard to life. This can be found specifically in Chapter 1, Administration, of NFPA 99. Therefore, it is be important to study the edition that was enforced at the time of construction as well.
The post "Back to basics: Medical gas storage under NFPA 99" appeared first on Consulting-Specifying Engineer

